Resources: Posters
A High-Resolution Multimodal Platform for Decoding Enteric Neuroimmune Interactions in Inflammatory Bowel Disease
September 5, 2025
Cell Symposia: Neuro-immune axis - Charting the periphery
Elisabet Rosas Canyelles, Ph.D.
Keywords: R3200 Platform, CellCage™ technology, Enteric nervous system, IBD (Inflammatory Bowel Disease), Neuroimmunology, Single-cell transcriptomics, Longitudinal analysis

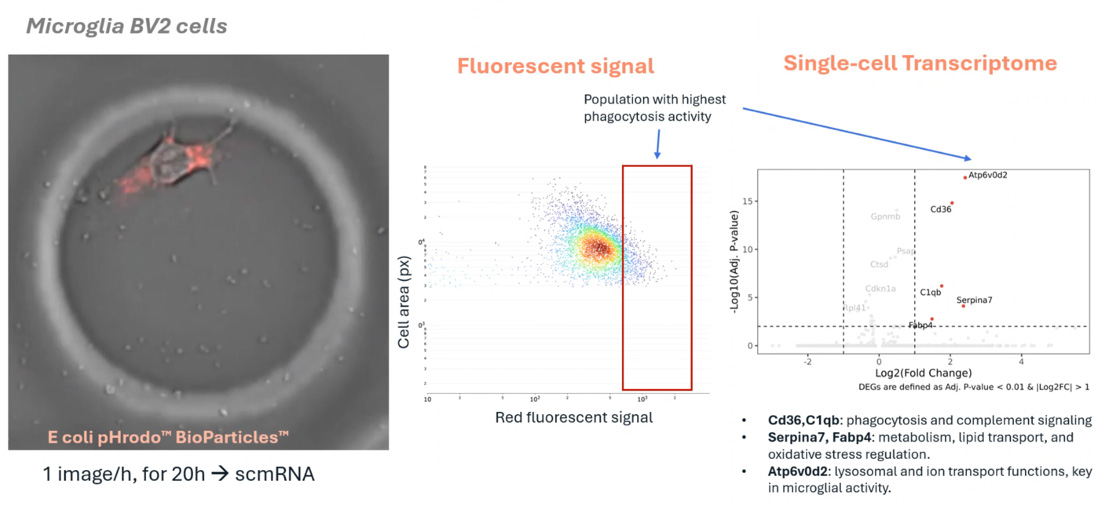
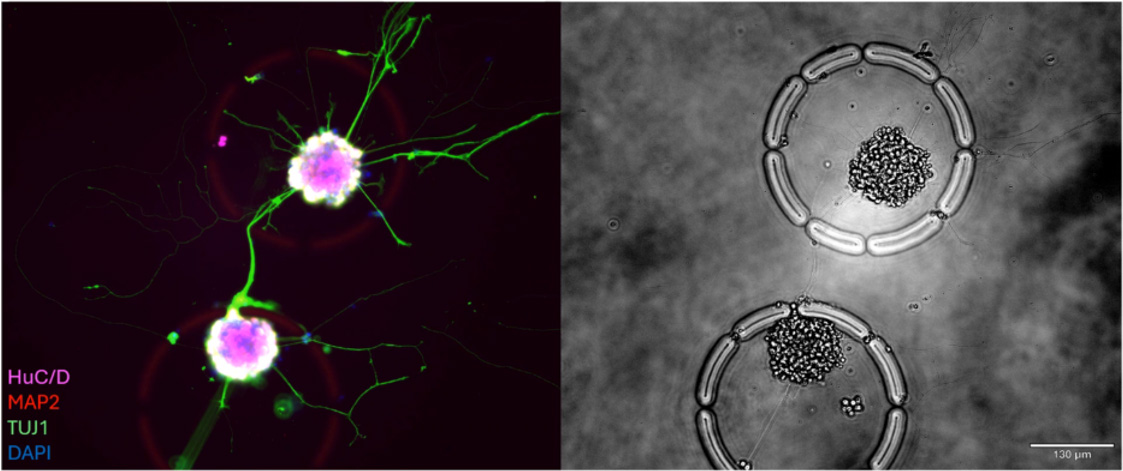
This research introduces a high-resolution multimodal workflow designed to decode complex enteric neuroimmune interactions in Inflammatory Bowel Disease (IBD). Utilizing the R3200 Platform and CellCage™ technology, the study captures longitudinal functional data and transcriptomic profiles from individual enteric neurons and immune cells. By preserving the spatial and functional context of these delicate cell types, the platform provides new insights into the cellular crosstalk driving intestinal inflammation and disease progression.
FAQ's
How does the R3200 Platform facilitate the study of enteric neuroimmune interactions?
The R3200 Platform enables the simultaneous, longitudinal monitoring of enteric neurons and immune cells within the same microenvironment. By capturing real-time interactions and physiological changes, the platform allows researchers to map the functional signaling pathways that occur between the nervous and immune systems in the gut.
What role does CellCage™ technology play in decoding IBD mechanisms?
CellCage™ technology provides a stable, non-destructive environment for sensitive enteric cell types. This allows for the precise tracking of cell-cell interactions over time without the stress of traditional dissociation, ensuring that the resulting transcriptomic data reflects the true physiological state of the cells during an inflammatory response.
Why is a multimodal approach necessary for studying the neuro-immune axis in IBD?
IBD is characterized by complex interactions that cannot be fully understood through gene expression alone. A multimodal approach on the R3200 Platform combines visual phenotyping—such as neuronal firing or cytokine release—with transcriptomics, providing a comprehensive view of how neuroimmune crosstalk contributes to intestinal dysfunction.