Resources: Posters
Concurrent live-imaging and single-cell transcriptome analysis of intact functional neuronal networks on a highly parallel scale
April 1, 2025
GRC | Gordon Research Conference: Functional Neurogenomics
Olaia F Vila, Ph.D.
Keywords: Neuronal networks, Functional neurobiology, Live-cell imaging, Single-cell transcriptomics, Synaptic plasticity, R3200 Platform, CellCage™ technology
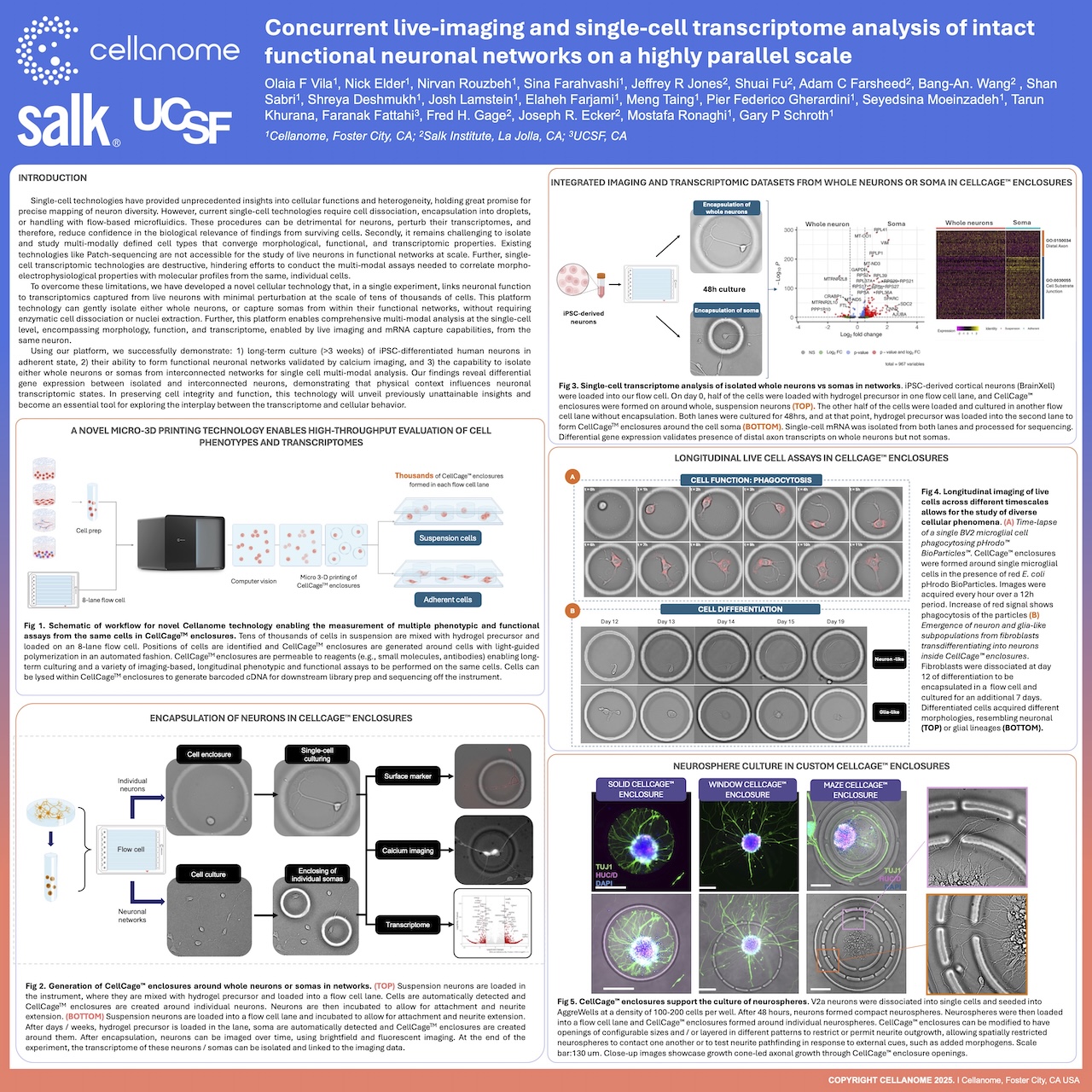
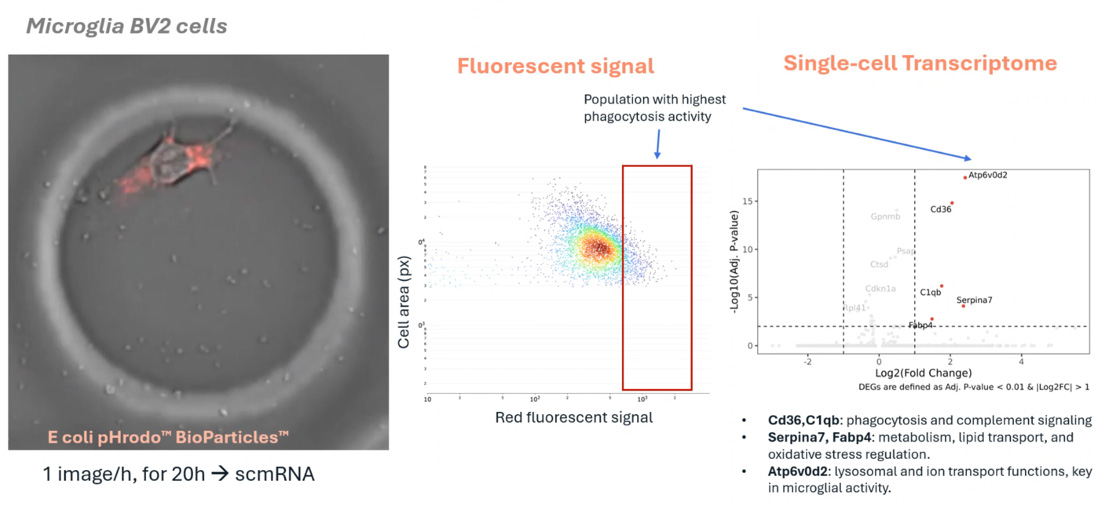
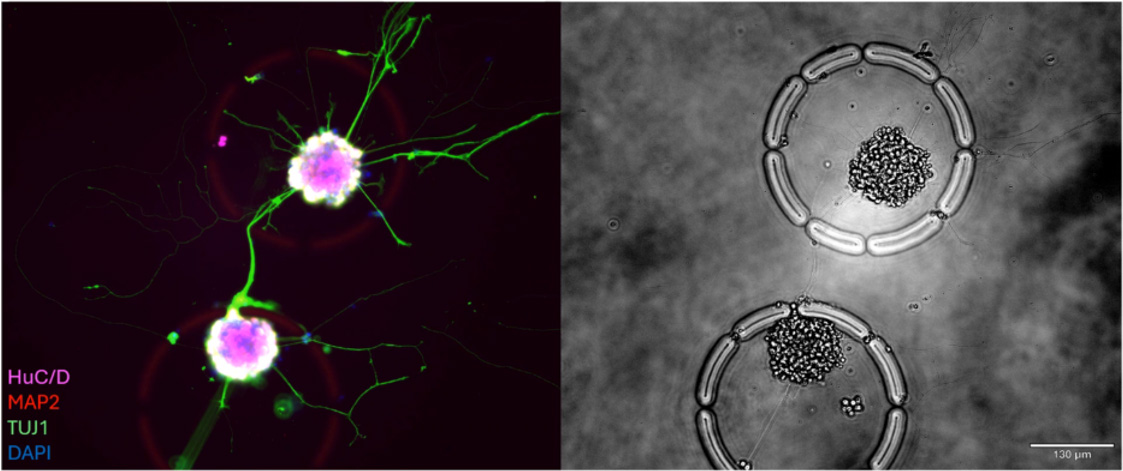
Leveraging the R3200 Platform and CellCage™ technology, this research enables the concurrent live-imaging and transcriptomic analysis of intact, functional neuronal networks. By maintaining the structural integrity of these networks on a highly parallel scale, the study reveals how complex cell-cell interactions and connectivity patterns correlate with underlying gene expression. This multimodal approach provides a powerful new framework for studying neurodevelopment, synaptic plasticity, and disease mechanisms in a preserved physiological context.
FAQ's
How does the R3200 Platform support the study of intact neuronal networks?
The R3200 Platform utilizes a highly parallel architecture to monitor multiple neuronal networks simultaneously. By capturing high-resolution, longitudinal imaging data across these networks before moving to sequencing, the platform ensures that the functional connectivity and spatial orientation of the cells are fully documented and linked to their individual transcriptomes.
What is the benefit of "concurrent" imaging and transcriptome analysis in neuroscience?
Concurrent analysis allows researchers to link real-time physiological activity, such as synchronized firing patterns or network-level signaling, directly to the molecular state of the participating cells. This eliminates the need for inference and provides a definitive map of the gene programs driving specific network behaviors.
How does CellCage™ technology preserve functional neuronal networks?
CellCage™ technology provides a non-destructive microenvironment that secures neurons and their established connections in place. Unlike traditional single-cell methods that require dissociation—which severs axons and dendrites—CellCage™ enclosures protect the intact network, allowing for the study of complex cellular architectures without sacrificing molecular resolution.