Resources: Posters
Identifying subsets of Senescent Cells using a Platform that Converges Key Hallmarks of Senescence to the Same Single Cells
October 20, 2025
Biomarkers of Aging
Annarita Scaramozza, Ph.D., Staff Scientist
Keywords: R3200 Platform, CellCage™ technology, Cellular senescence, Aging biomarkers, Senescence hallmarks, Multi-modal single-cell analysis, Metabolic disease
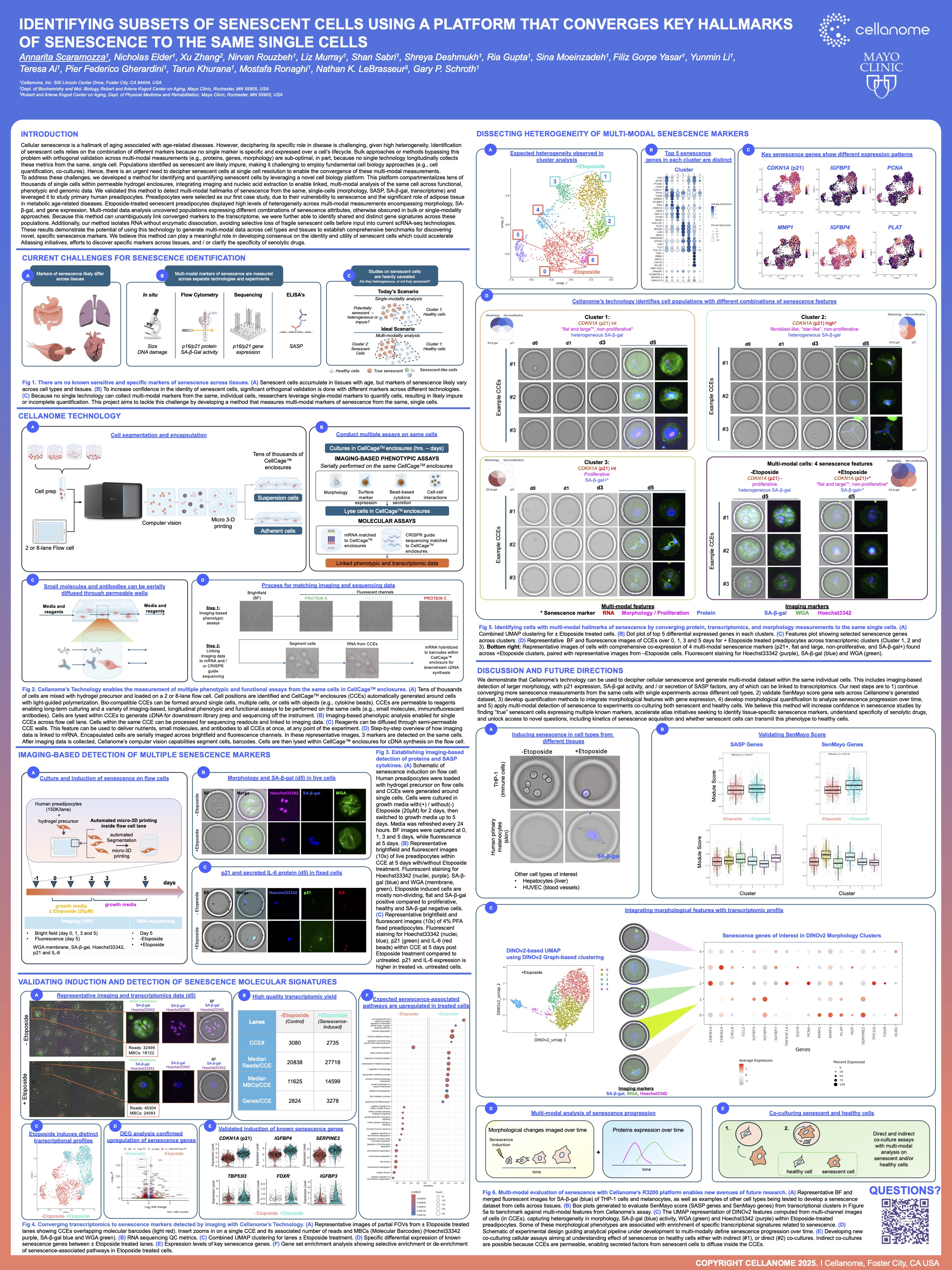
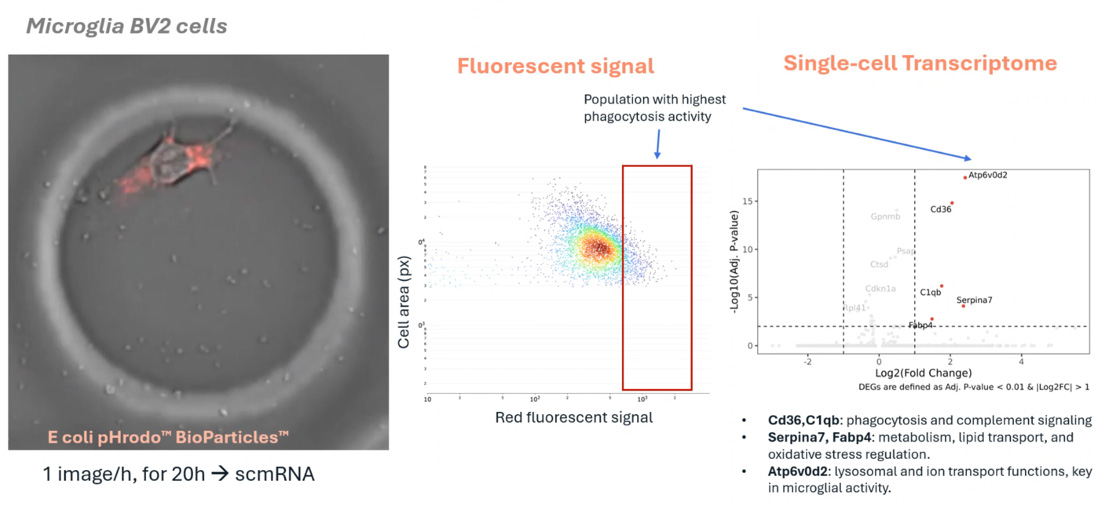
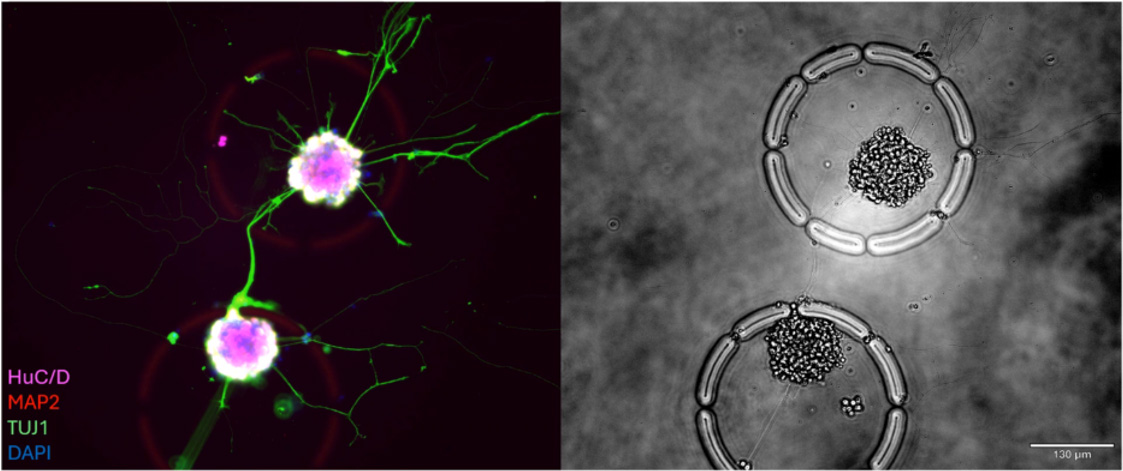
This study demonstrates a powerful approach to identifying distinct subsets of senescent cells by converging multiple hallmarks of senescence—such as morphology, secretory activity, and gene expression—to the same individual cells. Utilizing the R3200 Platform and CellCage™ technology, the research overcomes the limitations of single-marker identification, providing a high-resolution, multimodal map of cellular aging. The results establish a new standard for characterizing senescent heterogeneity in aging and metabolic disease research.
FAQ's
How does the R3200 Platform help identify subsets of senescent cells?
The R3200 Platform enables the longitudinal tracking of individual cells, allowing researchers to observe multiple senescence hallmarks simultaneously. By linking real-time functional data with end-point transcriptomics through CellCage™ technology, the platform can distinguish between various senescent phenotypes that appear identical under traditional single-marker assays.
Why is it important to converge "hallmarks of senescence" to the same single cells?
Senescence is a complex, multi-stage process with no single universal marker. Converging hallmarks like morphology, lysosomal activity, and RNA expression to the same cell ensures that identifying a "senescent" state is biologically accurate and allows for the discovery of rare or highly specific cellular subsets involved in aging.
How does CellCage™ technology improve the study of cellular senescence?
CellCage™ technology secures cells in a stable microenvironment, preventing the loss of delicate senescent phenotypes during the analysis process. This non-destructive approach allows for the continuous monitoring of the Senescence-Associated Secretory Phenotype (SASP) and other dynamic behaviors before moving to high-resolution sequencing.