Resources: Posters
Platform for Large-Scale Analysis of Brain Cell Types at Single-Cell Resolution Spanning Longitudinal Multi-Modal Measurements
November 19, 2025
Society for Neuroscience 2025
Olaia Villa, Ph.D., Assoc. Director, Collaborations
Keywords: R3200 Platform, CellCage™ technology, Neurobiology, Neural networks, Brain cell types, Longitudinal multi-modal analysis, Single-cell resolution
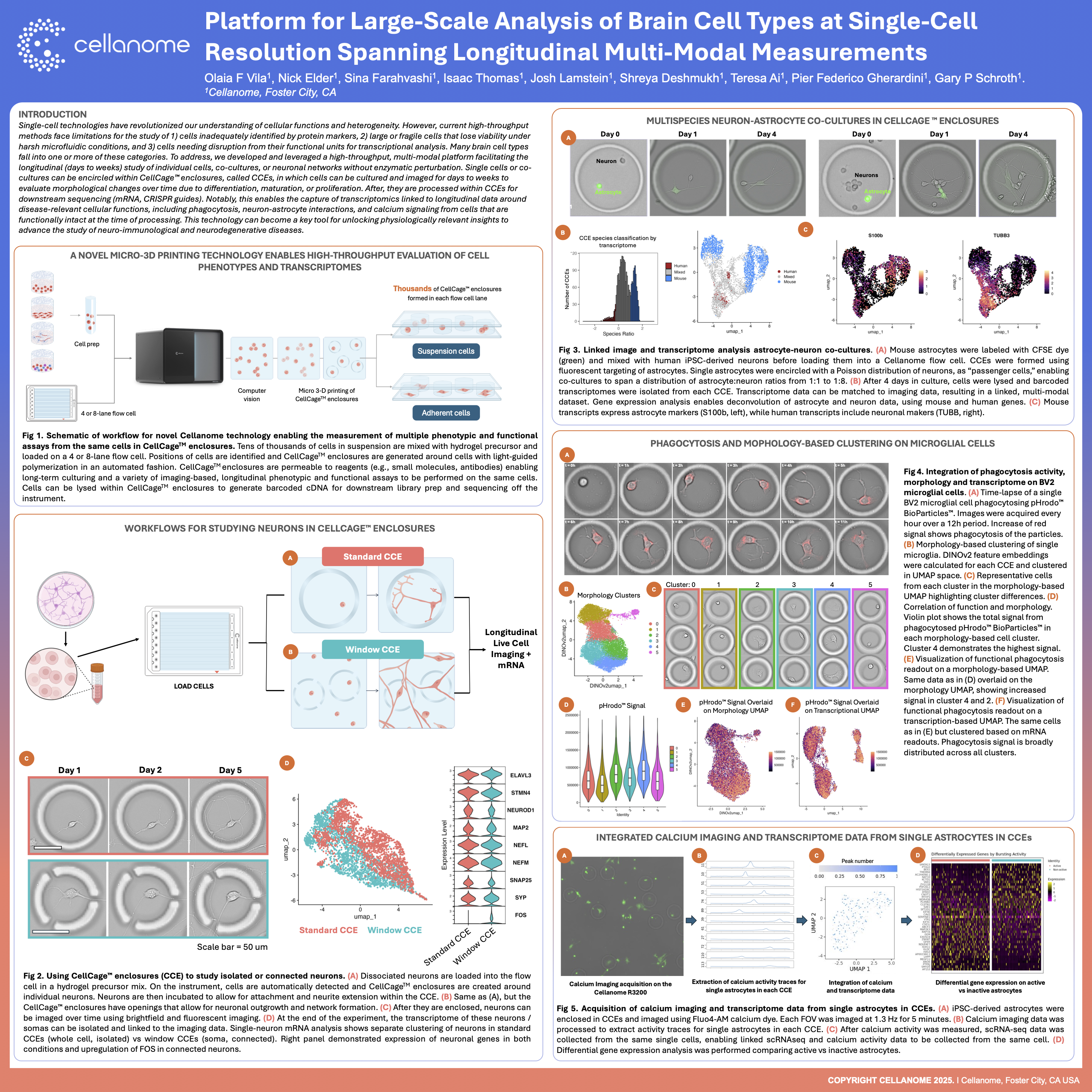
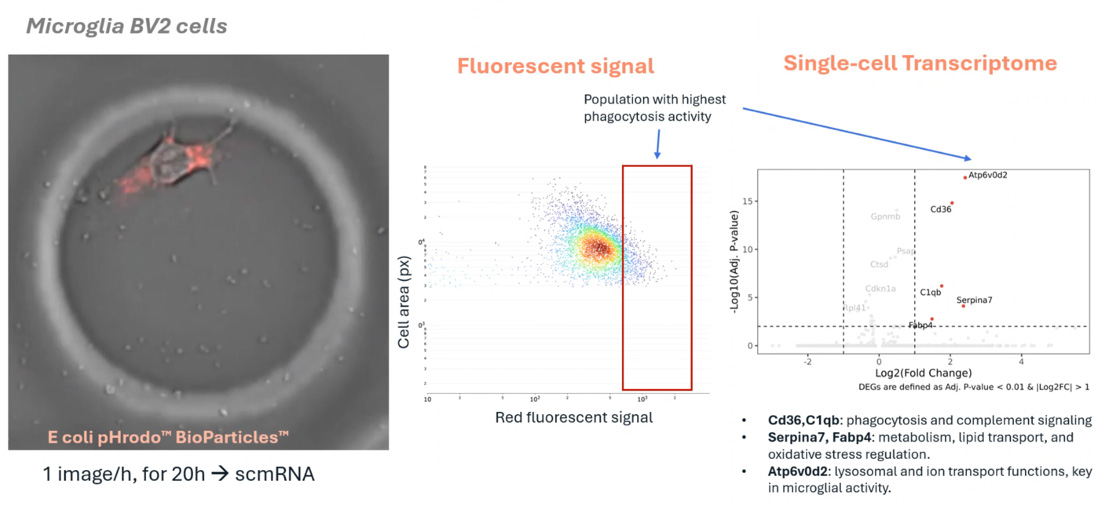
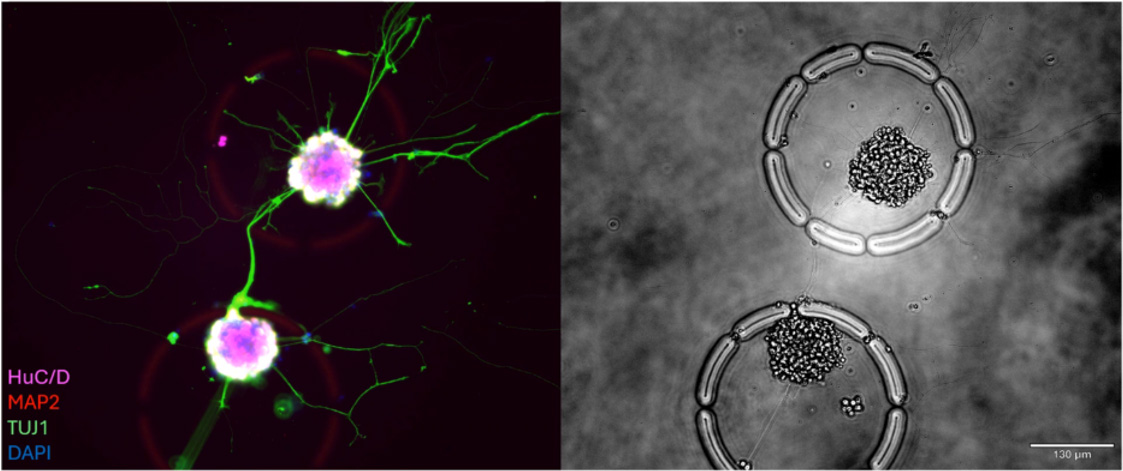
This study introduces a high-throughput workflow for the large-scale analysis of brain cell types, utilizing the R3200 Platform and CellCage™ technology. By spanning longitudinal multi-modal measurements, the research enables the precise tracking of neural networks and individual neuronal behaviors over time. The integration of advanced live-cell imaging with single-cell transcriptomics provides a comprehensive framework for studying brain function and disease states within controlled, tissue-like environments.
FAQ's
How does the R3200 Platform facilitate large-scale analysis of brain cells?
The R3200 Platform utilizes a highly parallel architecture that allows for the simultaneous monitoring of hundreds of individual brain cells or small neural clusters. This scalability enables researchers to conduct high-throughput studies of complex brain cell types while maintaining single-cell resolution across every sample.
What are the benefits of longitudinal multi-modal measurements in neuroscience?
Longitudinal measurements allow for the observation of dynamic neural behaviors, such as axon extension or synapse formation, as they unfold. By combining these temporal observations with end-point transcriptomics through CellCage™ technology, researchers can link specific physiological developmental trajectories to underlying genetic drivers.
How does CellCage™ technology support the study of neural networks?
CellCage™ technology provides a stable microenvironment that secures brain cells in place without traditional tissue dissociation. This preserves the structural integrity of neural networks and allows for the precise study of cell-cell interactions and long-term functional changes in a setting that more closely mimics native brain tissue.