Resources: Posters
Unveiling genes in dendritic cells that enhance T cell priming through functional multiplexed single cell-cell interaction analysis
April 12, 2026
2nd Annual ARC Symposium 2026/ SITC 2025
Camilla Valente, Ph.D., UCSF
Keywords: Dendritic cells, T cell priming, CRISPR screening, CellCage technology, single-cell analysis, immunosuppression, immunotherapy, Cellanome R3200.

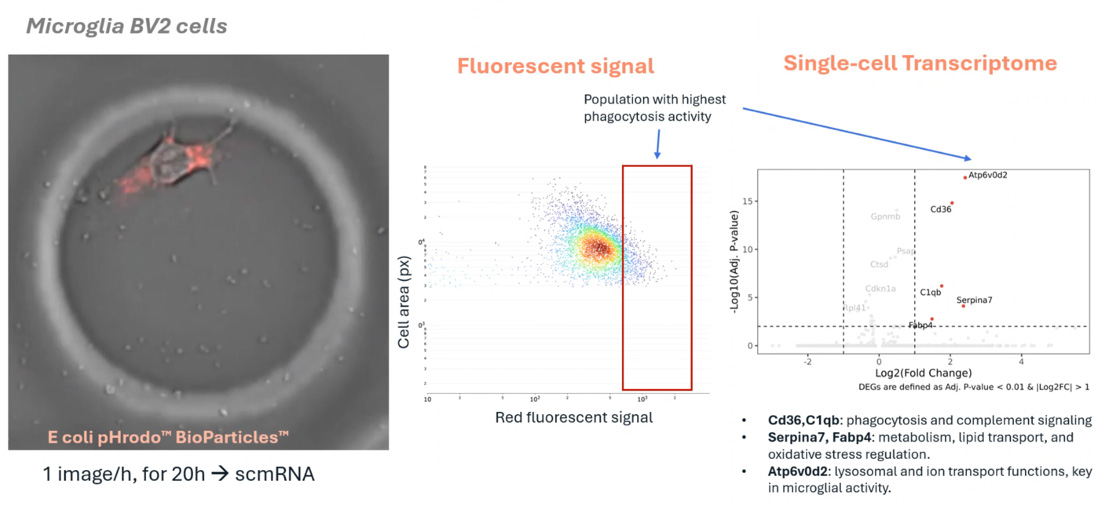
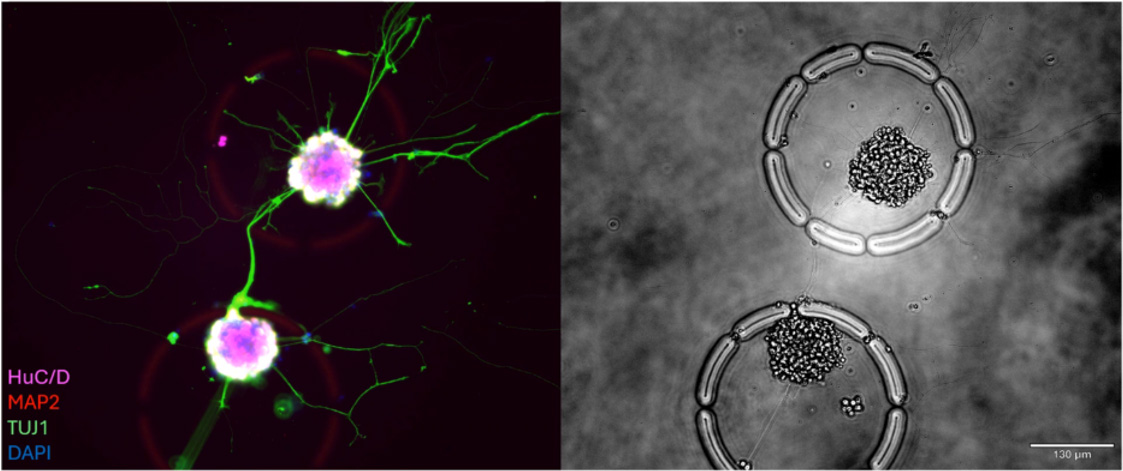
In this poster, Camilla Valente (UCSF), details a novel pooled CRISPR screening workflow to identify genetic regulators of dendritic cell (DC) function. Using the Cellanome R3200 Platform, researchers enclosed single edited DCs with T cells in CellCage™ enclosures (CCEs) to monitor priming dynamics via time-lapse imaging. The study successfully links specific genomic perturbations to the rescue of T cell activation (CD69+ expression) in DCs conditioned with immunosuppressive factors like IL-10 and TGF-beta
Key Takeaways
- Functional CRISPR Mapping: Directly links specific gene knockouts in dendritic cells to functional T cell outcomes (proliferation and activation) at a single-cell-pair level.
- Overcoming Suppression: Identifies genetic targets that allow DCs to resist cancer-driven immunosuppression and successfully prime OT-I T cells.
- High-Throughput Imaging: Utilizes longitudinal imaging to capture the "cellular choreography" of immune synapse formation and activation markers over 3 days.
FAQ's
What conditions were used to simulate the immunosuppressive tumor microenvironment in this study?
Researchers conditioned bone-marrow-derived dendritic cells (BMDCs) with IL-10 and TGF-beta to model the suppressive signaling typical of a tumor environment. This conditioning was shown to significantly impair the DCs' ability to prime T cells—reducing CD69 expression and proliferation—which established the baseline for testing CRISPR-based genetic rescues on the Cellanome R3200 platform.
What markers were used to define successful T cell priming?
Priming success was quantified by tracking T cell proliferation and the upregulation of the early activation marker CD69 through longitudinal fluorescent imaging within the CellCage™ enclosures.
What is the advantage of using CellCage™ (CCE) technology for this screen?
CCEs allow for the isolation of single DC-T cell interactions in a biocompatible, semi-permeable environment. This prevents the "averaging" effect of bulk co-cultures and enables researchers to link a specific CRISPR edit in one DC to the exact functional response of the T cells it interacted with.