Resources: Posters
A Single-Cell Platform for Longitudinal Monitoring of Protoplast Physiology and Transcriptomics
April 12, 2026
AGBT Agriculture Meeting
James Walker, Ph.D., The Salk Institute for Biological Studies
Keywords: Plant biology, AgBio, Protoplast development, Stress response, Single-cell plant genomics

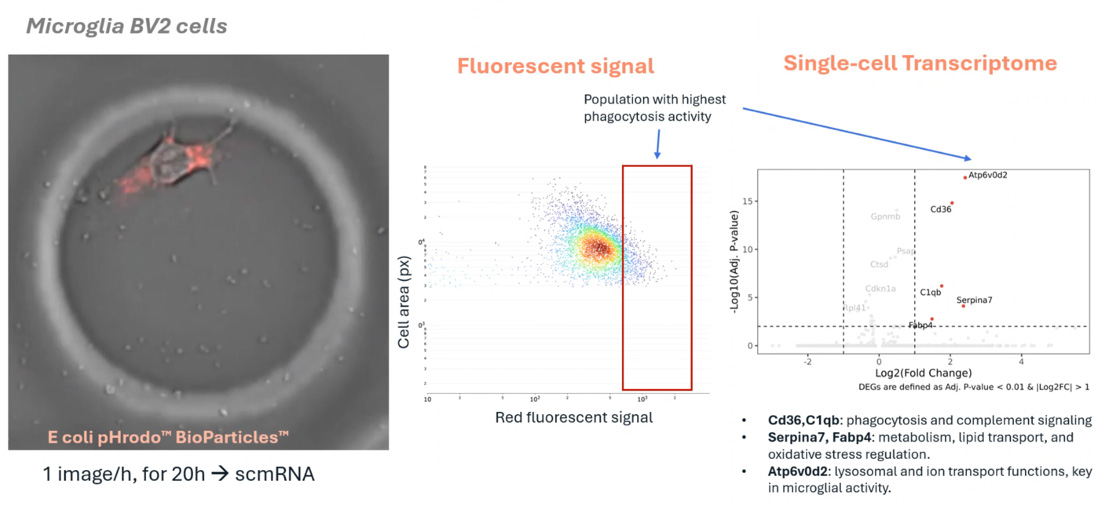
Highlighting a novel workflow for plant functional genomics, this poster demonstrates the longitudinal monitoring of intact protoplast physiology and transcriptomics. By preserving cell health in a controlled microenvironment, the platform reveals dynamic developmental trajectories and physiological changes at single-cell resolution, avoiding the stress-induced artifacts of traditional tissue dissociation.
FAQ's
Why is longitudinal monitoring essential for protoplast research?
Protoplasts are highly sensitive to environmental changes. Longitudinal monitoring enables the observation of real-time physiological shifts and developmental transitions, providing a dynamic view of plant cell health and response that single-point assays cannot provide.
How does the platform improve plant transcriptomic studies?
Traditional plant transcriptomics often requires tissue dissociation, which can induce stress and alter cellular data. This platform enables the study of intact protoplasts in a controlled environment, linking continuous physiological observations to high-resolution transcriptomic insights.